|
In general, atoms are most stable, least reactive, when their outermost electron shell. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons. By going through the periodic table, we see that the electron-dot symbol of atoms will never have more than eight dots around the atomic symbol. The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. Elements in the same group on the periodic table have the same number of valence electrons. When forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. A gap in ionization energies could tell us how many valence electrons an element has. 
Valence electrons are found in the s and p orbital of the outermost shell. The number of dots equals the number of valence electrons in the atom. Valence electrons are the outermost electrons in an atom. The last electron out of eleven goes to the. Sodium configuration is 1s 2 2s 2 2p 6 3s 1. It is an alkali metal that participates in the formation of bonds via one valence electron. What is the atomic number Scientist Henry Gwyn Jeffreys Moseley researched the X-ray spectrum of various elements in 1913-1914. Sodium is an alkali metal and its symbol is ‘Na’. chlorine, but once the elements form the compound sodium chloride (table salt). Therefore, the atomic number of sodium is 11. We use Lewis dot structures to map these valence electrons in order to. In methane, carbon has a valence of 4 in ammonia, nitrogen has a valence of 3 in water, oxygen has a valence of 2 and in hydrogen chloride, chlorine has a valence of 1. The sodium atom contains a total of eleven electrons and protons. Sodium (Na) is the eleventh element in the periodic table. The combining capacity, or affinity of an atom of a given element is determined by the number of hydrogen atoms that it combines with. With the next element, sodium, the process starts over with a single electron because sodium has a single electron in its highest-numbered shell, the n = 3 shell. A Lewis electron dot symbol (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Valence electrons are electrons found in the outermost shell of an atom. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. This is required by the law of conservation of matter as well.\] The electronegativity of Sodium is: 0.93 In general, an atom’s electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The need for the number of electrons lost being equal to the number of electrons gained explains why ionic compounds have the ratio of cations to anions that they do. These three ions attract each other to give an overall neutral-charged ionic compound, which we write as Na 2O. What we need is a second Na atom to donate a second electron to the O atom: Transition elements or transition metals. 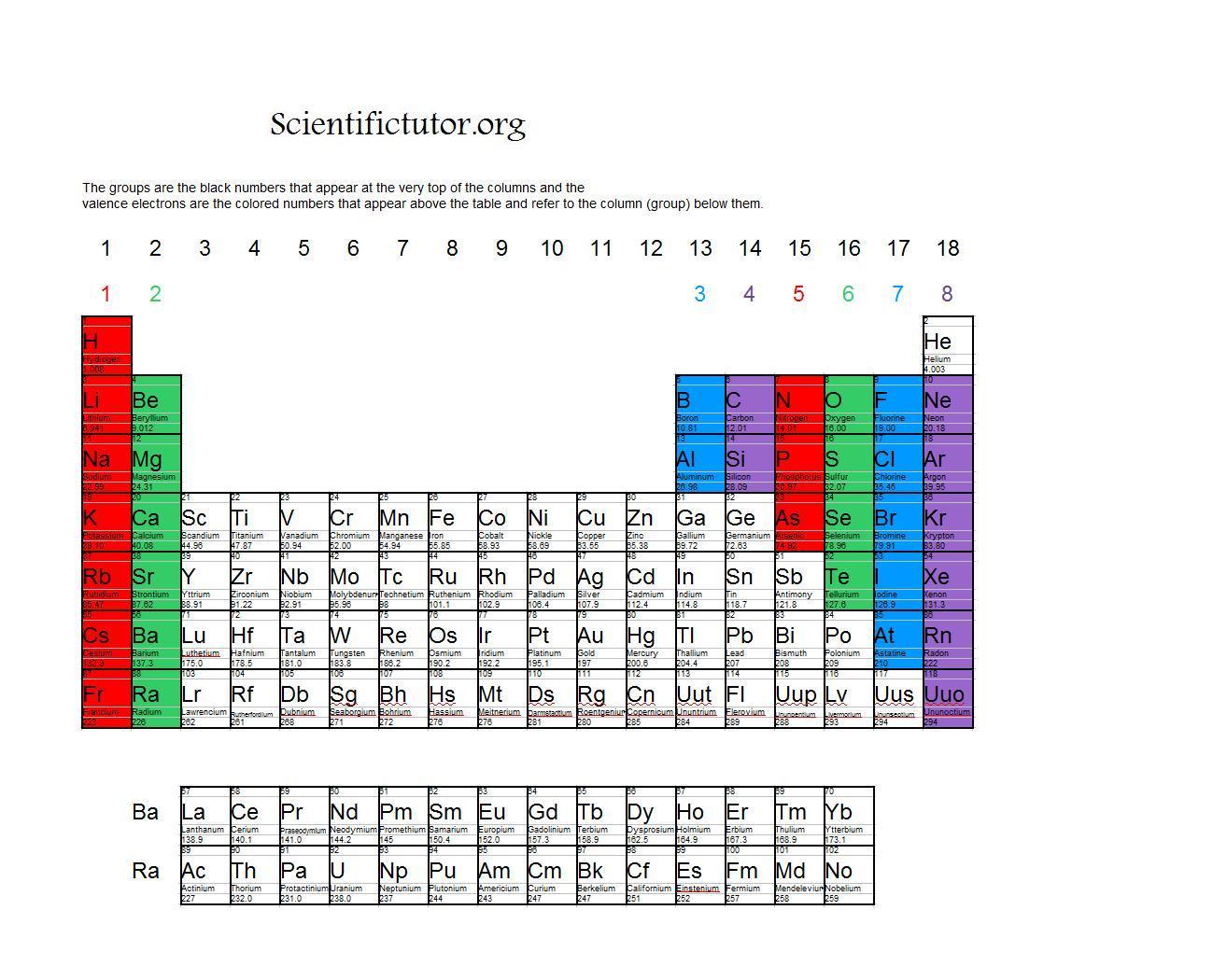
E) eight minus the group number., Valence electrons are electrons located A) in the outermost energy level of an atom. The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This electron arrangement indicates that the outermost orbit of Sodium (Na) has 1 electron. Hence sodium element has the electrons arrangement 2, 8, 1. The completely filled d orbitals count as core, not valence, electrons. Study with Quizlet and memorize flashcards containing terms like The number of valence electrons found in an atom of a Group A element is equal to A) its atomic number. Now the atomic number of Sodium (Na) is 11. For example, gallium (Ga, atomic number 31) has the electron configuration Ar4s 2 3d 10 4p 1, which contains three valence electrons (underlined). 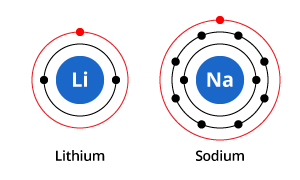
The O atom still does not have an octet of electrons. The valence electrons for main group elements are those with the highest n level.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
